Amazing artwork by:(http://browse.deviantart.com/art/music-106660536)
Amazing artwork by:(http://browse.deviantart.com/art/music-106660536)
One good thing about music, when it hits you, you feel no pain.
-Bob Marley
This is one of my most favourite quotes. Music is an important part of my life. Even though I may not be musically talented per say (does singing in the shower count?), I enjoy listening to it daily as it helps me forget about life’s problems instantly. For those few minutes I’m in a zone where nothing else matters except how the music makes me feel in that moment; be it happy, sad, calm you name it, music can take you on an emotional roller-coaster.
So when I read an article talking about assigning musical notes to each amino acid in different proteins to create a melody I was beyond thrilled!
This has been an ongoing project by Rie Takahashi and Jeffrey Miller, colleagues at the University of California at Los Angeles, in which their aim is to “musicalise” the amino acid sequences in various proteins. Although this idea has been thought of before, Takahashi and Miller have found a way to make the tunes richer and more rhythmic compared to earlier efforts that resulted in jumpy notes.
Rie Takahashi
Jeffrey H. Miller
So, how is this done?
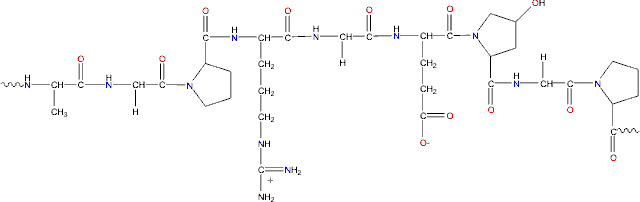
It’s really simple actually. Each of the 20 amino acids is allocated a specific musical note, be it middle C or D sharp, until each one has their own note. Then a protein is chosen and a musical score is made just by transcribing the amino acids of the protein’s sequence into musical notes.
Remember I told you that previous attempts at making “musical proteins” failed because of notes “jumpy notes”? This was because sometimes notes would leap 20 notes at a time, thus making it hard/ unmelodious to the ear. But Takahashi and Miller overcame this obstacle by giving each amino acid not just one note but three notes, also called a triad chord. These triad chords were played successively and the harmonies were easier on the ears and just overall nicer to listen to. They did cheat a little bit though because they made minor changes to the chords already used in the first 13 amino acids and then gave them to the remaining 7, but it was all to make the highest tunes more favourable.
They also discovered a way to put in timings to each triad so rhythm could be introduced into the music. By using the changes in the codon (triplets of DNA bases in the gene) frequencies, they allotted time values to the chords for each of the amino acids in the protein sequence. Since an amino acid can have up to 4 different codons, the more common the codon is in the DNA, the longer the time value it has. That means the longest note in the melody would have a semibreve enduring 4 beats! Cool right?
I’m sure you just want to hear what it sounds like by now, so listen here!! On their website, they have numerous other creations here. Even though it’s not Beethoven, I still think it’s an ingenious idea and I’m excited to see and hear what becomes of this as Takahashi would like to add other instruments to the protein music as well!
Other vids can be found on Youtube as well showing how others convert each amino acid sequence of different proteins into music 🙂
Article Reference: Coghlan, Andy. 2007. “Music made to measure from nature’s proteins” Accessed April 6, 2013. http://www.newscientist.com/article/dn11775-music-made-to-measure-from-natures-proteins.html
References:
http://www.mimg.ucla.edu/faculty/miller_jh/gene2music/home.html
http://www.newscientist.com/article/dn11775-music-made-to-measure-from-natures-proteins.html